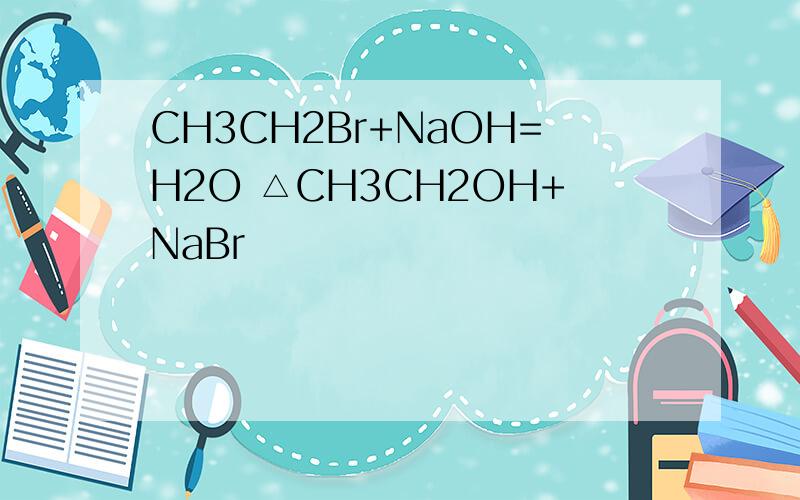
CH3CH2Br+NaOH=H2O △CH3CH2OH+NaBr
CH3CH2Br+NaOH=H2O △CH3CH2OH+NaBr
CH3CH2OH+HBr→CH3CH2Br+H2O
Br2+2NaOH=NaBr+NaBrO+H2O (或3Br2+6NaOH=5NaBr+NaBrO3+3H2O)
ch3ch2oh+h2o=什么?
CH3CH2BR与CH3CH2OH的熔沸点
NaOH+?H2O=NaOH*?H2O
为什么可以用NaOH来除去溴苯中的溴,Br2 + 2NaOH = NaBr + NaBrO + H2O里的NaBrO怎么
化合价升降法配平~Br2 + NaOH = NaBr + NaBrO + H2O像这种Br的歧化反应,该怎么考虑化合价升
“双线桥”法配平:NaSx+NaBrO3+NaOH----Na2SO4+NaBr+H2O.最后附有说明)
此方程式的原理?3Br2+6NaOH→5NaBr+NaBrO3+3H2O
2 CH3CH2OH + O2 == 2 CH3CHO + 2 H2O
对于这个可逆反应2CO+4H2=CH3CH2OH+H2O